Alzheimer’s disease (AD) is the leading cause of dementia affecting over 25 million people worldwide. It is expected to rise to over 81 million people by 2040 (Ferri et al., 2005). Currently, someone develops AD every 68 seconds in America (Thies et al., 2013). The most common early symptoms include loss of short-term memory, irritability, and confusion, which can worsen over time leading to loss of long-term memory and inability to care for oneself (Waldemar et al., 2007). Currently there is no cure for AD and new aspects of the disease are being discovered every day. AD is a neurodegenerative disease that results in neuropathological changes, including neuronal loss and gross atrophy of affected regions in the brain (Barber, 2012). These neuropathological changes are due to the extracellular accumulation of plaques composed of the peptide amyloid-beta (Aβ) as well as accumulation of intracellular neurofibrillary tangles (NFTs) (Hardy and Selkoe, 2002) that interrupt neuronal communication and eventually lead to neuronal death.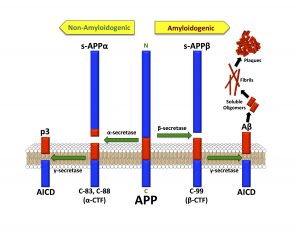
These amyloid-beta plaques arise from cleavage of Amyloid Precursor Protein (APP). Many cellular enzymes act on APP, but only the actions of β-secretase and γ-secretase have been shown to result in Aβ production (Chow et al., 2010). β-secretase cleaves APP to produce s-APP-β and CTF99 or CTF89, depending on its carboxy-terminal position, and is mediated by BACE1. γ-secretase contains four proteins with protease activity that cleave APP in its intra-membrane domain, resulting in Aβ, p3, or AICD59/57. The resulting Aβ peptide can range from 38 to 43 amino acids. The most amyloidogenic are Aβ 1-42 and 1-40 (Aβ42 and Aβ40, respectively) (Chow et al., 2010). The Aβ peptides are cleaved from APP on the cell surface, or inside endocytotic vesicles, and are then released into the extracellular space where oligomerization occurs and plaques are formed.